HEOR
Select a fit-for-purpose health economics and outcomes (HEOR) consulting partner
We ensure optimal value demonstration for innovative health technologies through a uniquely flexible, end-to-end HEOR consulting team solution powered by therapeutic knowledge, tech-enabled insights, and methodological expertise.

Our evidence solutions include:
- HEOR, value and access (including pricing)
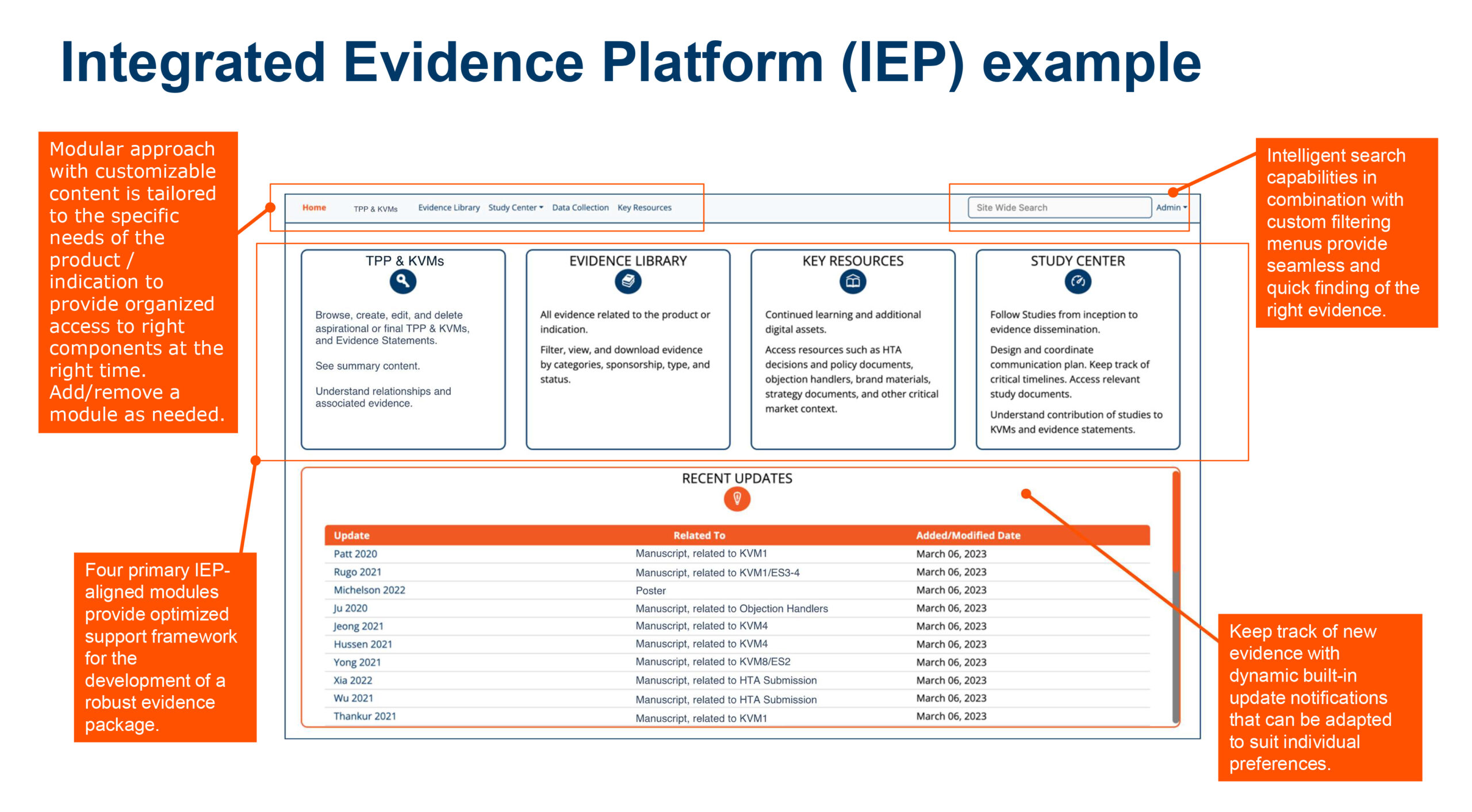
- Integrated evidence planning (IEP)
- Value proposition/value story/evidence gap analysis
- HEOR/HTA advisory boards
- Appraisal review/HTA/scientific advice
- Outcomes-based contracting
Landscape
- Competitive landscape
- Pricing and reimbursement landscape assessment
- Analog assessment
- PRO landscape
- Policy landscape
Generation
- Rapid evidence synthesis/Disease state profiling
- Disease model
- Economic model (BIM, CEM, Breakeven model)
- Cost calculator
- Global patient funnel
- Model adaptations
- Meta analysis and indirect comparisons
- Preference survey
Communication
- Value dossiers (GVD, AMCP)
- Tool and resources for customer-facing teams (MSL, HOLs, AM)
- Response letter and submissions to HTAs
- Objection handlers
- Access playbook
- Integrated Evidence Generation Planning playbook
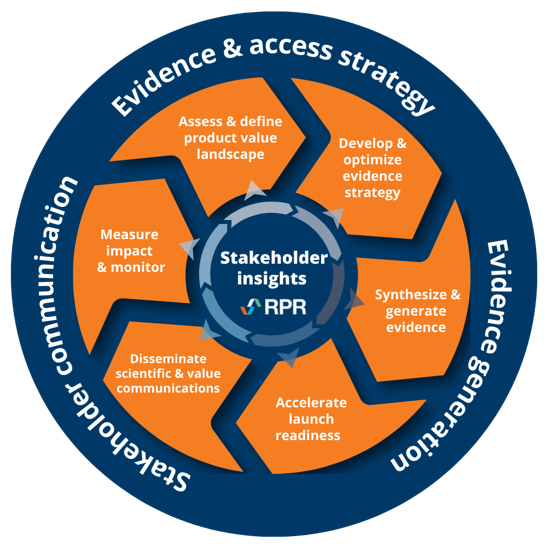
The power of an integrated approach
Leveraging extensive methodological expertise, tech-enabled insights, and experience across indications, we help you plan strategically, and generate impactful evidence. Our end-to-end HEOR solutions – from sophisticated modeling to AI-powered evidence synthesis, and HTA support – deliver the impactful value demonstration you need to help your products reach their full potential.
Led by professionals with experience as both HEOR consultants and within pharma, biotech, and medical device organizations, the depth and breadth of our expertise enables us to offer a one-stop solution for all your health economics and outcomes research consulting needs.
- Fit-for-purpose strategies resulting in impactful evidence
- Innovative Flexible Integrated Team (FIT) engagements
- Rapid stakeholder engagement to optimize both strategy and messaging
- Enhanced communication, evidence synthesis, analytics, modeling, access and pricing, and value demonstration
- Evidence generation planning and transparent evidence management platforms
- Data agnostic real-world evidence expertise
“We are impressed with the efficiency and quality of the work, your leadership skills, and the extra efforts you've made to ensure the results are absolutely accurate."
HEOR/RWE client
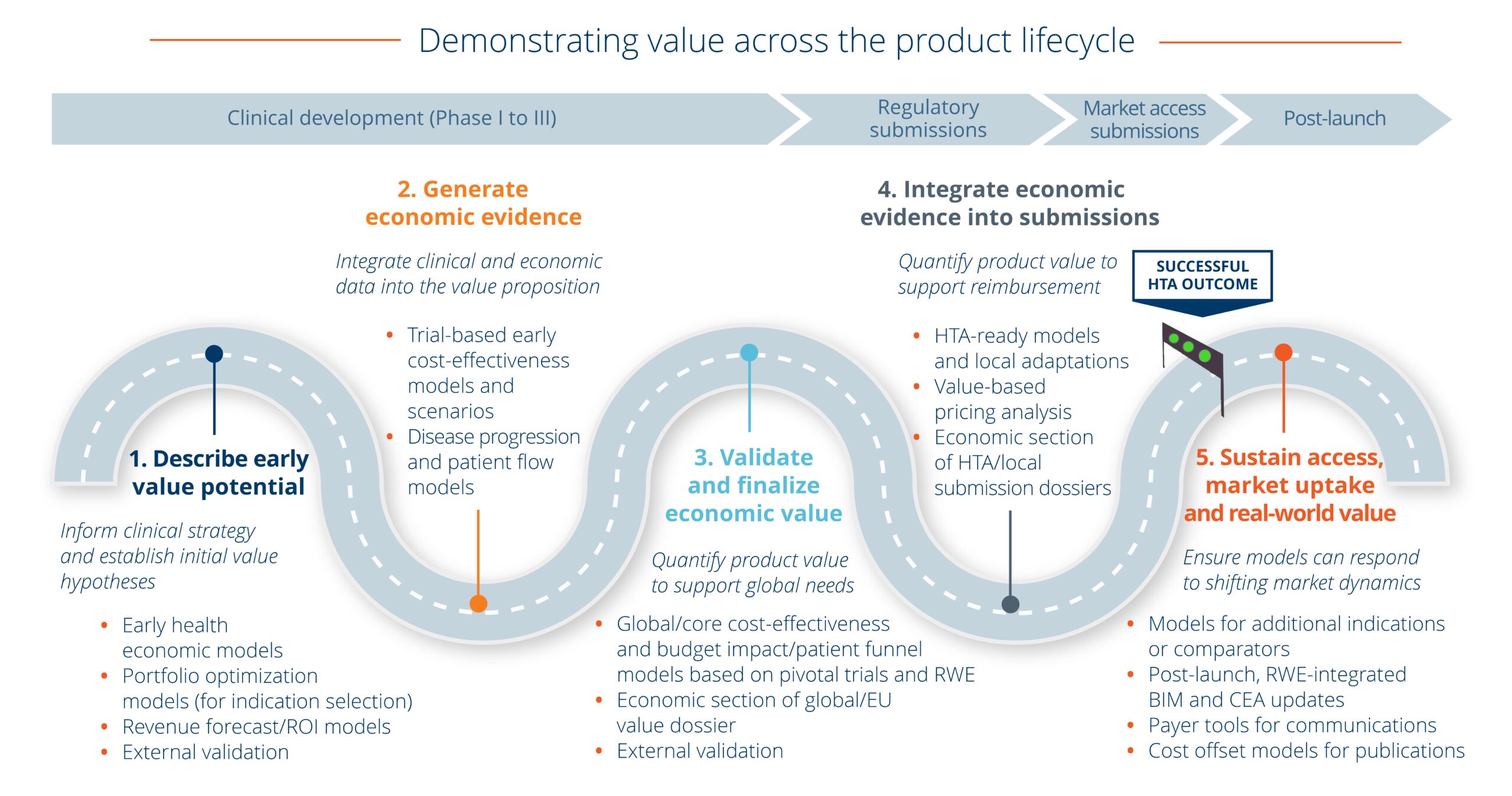
Economic modeling: A strategic roadmap
We have proven expertise in the design and development of all types of economic models. From early value and global cost-effectiveness models through HTA-ready submission models to post-launch stakeholder communication model tools, we support your team to optimize evidence-based pricing, shape reimbursement strategy, accelerate adoption, and unlock market access across the product lifecycle.
It’s in our DNA. Discover your new strategic partner
Our strategic consulting framework provides a clear path from evidence to impact. We can provide you with a robust framework for strategic thinking, unbiased perspectives, clear next steps/rationale, and confidence in your decision-making.
Therapeutic areas
Our team possesses deep knowledge across therapeutic areas, further details of which as can be explored through our selected publications list.
- Autoimmune
- Cardiology
- Dermatology
- Diabetes
- Endocrinology
- Gastroenterology
- General medicine
- Hematology
- Hematology/oncology
- Immunology
- Infectious diseases
- Mental health
- Musculoskeletal
- Nephrology/ Renal disease
- Neurology
- Obesity
- Obstetrics/ Gynecology
- Oncology
- Ophthalmology
- Pain
- Psychiatry
- Pulmonology/ Respiratory
- Radiology
- Rheumatology
- Urology
Looking for a comprehensive solution to your health economics and outcomes research challenges?
Our 8-step integrated evidence generation plan framework
Explore our wider integrated evidence generation planning offering: meeting the needs of multiple stakeholders across geographies and through the full product lifecycle.



Flexible Integrated Teams (FIT)
Research partnerships reimagined
Many clients seeking HEOR support choose to take advantage of our optional FIT engagement model, which provides tailored, cross-functional teams that integrate seamlessly within your own to fill experience gaps or supplement existing resources.
The size and composition of your team can gradually evolve or quickly pivot to meet your changing needs, all without the costly delays involved in adjusting a statement of work.
A trusted extension of your organization, our team becomes your team.
Accelerated Insight Generation
Where agile technology meets HEOR expertise
Our expert HEOR consultant teams leverage a suite of technology platforms to optimize evidence generation and communication.
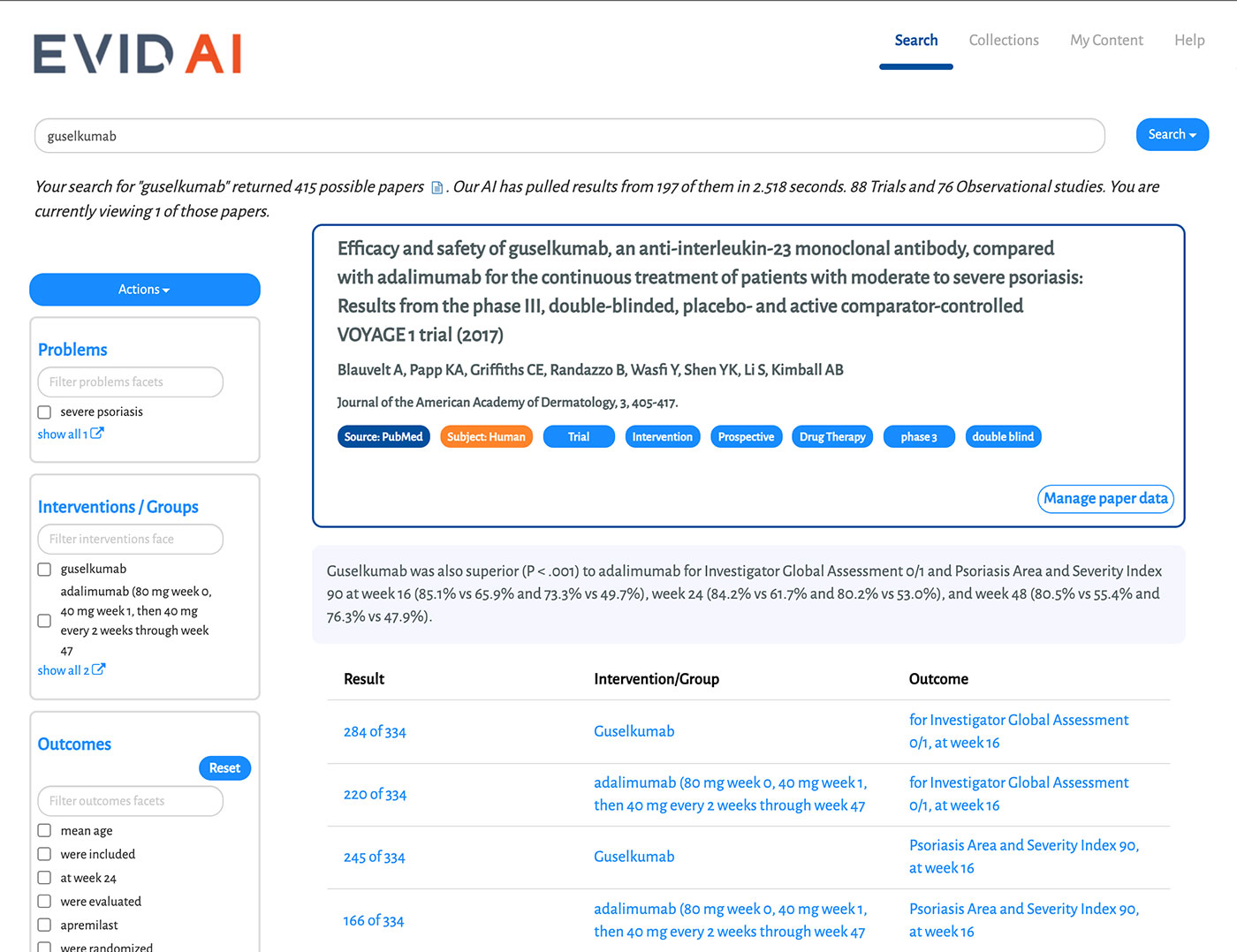
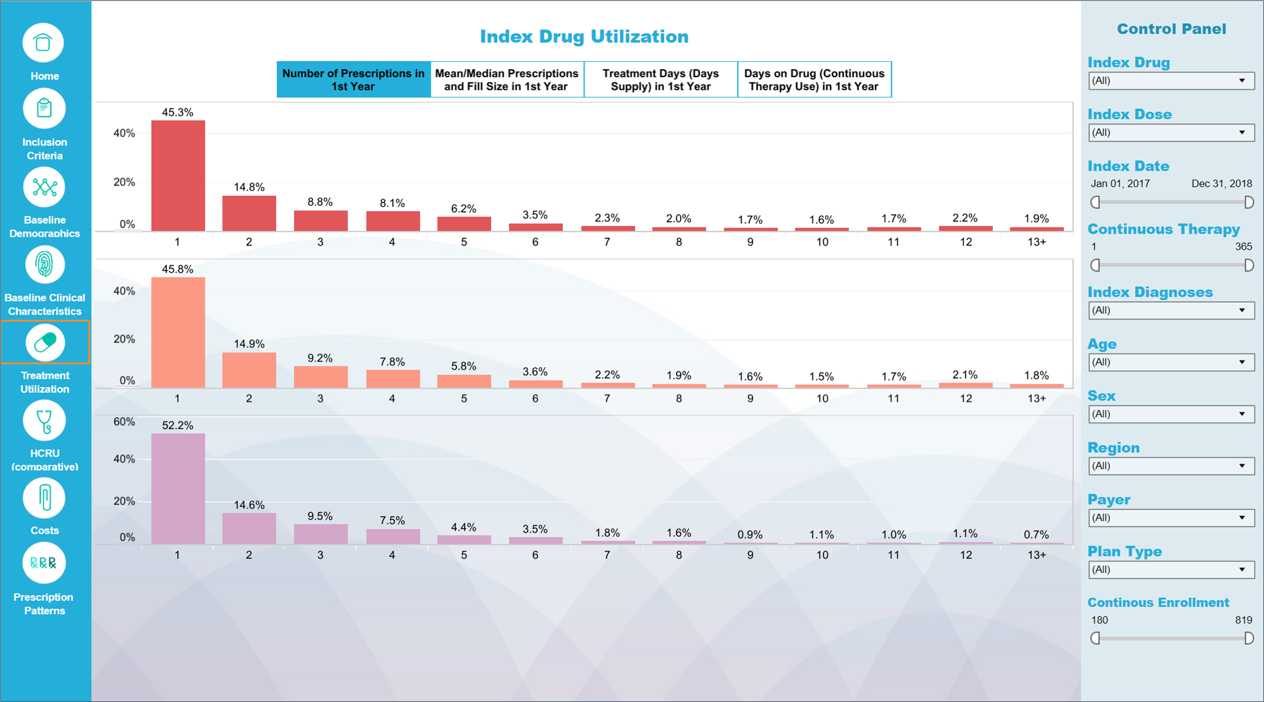
EVID AI enhances and accelerates literature reviews; RPR secures robust stakeholder insights at any stage of the process; and our interactive dashboards transform complex data into digestible insights.
Additionally, we have developed Integrated Evidence Planning platforms to meet the challenge of evidence organization and communication, providing optimal visibility to internal stakeholders and ensuring proper alignment across functional and geographic teams.
Tech-enabled HEOR
Rapid stakeholder insights


Technology-infused research
Trust the voice of the stakeholder to test your evidence strategy and optimize market access
High quality engagement with payers and other stakeholders can significantly enhance decision-making at all stages of product development. We are able to provide clients with both a global, vetted stakeholder network and a transparent online platform to facilitate this engagement.
- Leverage our agile, tech-enabled RPR platform to develop robust market access strategies and fit-for-purpose evidence-generation plans.
- Secure critical payer, key opinion leader (KOL), patient advocacy group, and policy maker insights across the product lifecycle.
- Engage with a global network of 3,500+ vetted stakeholders across 65+ countries to test your market access and evidence strategies as they evolve.
HEOR Case Study
Focal segmental glomerulosclerosis: evaluate HCRU and burden of illness
Focal segmental glomerulosclerosis (FSGS) is a varied, complex pattern of kidney damage that has several different causes and numerous names and terminology, as well as different proposed classification systems.
Evidence generation required (1.) claims to summarize the cost and HCRU burden and (2.) clinical data (e.g. labs from EHR) to accurately identify patients in the study cohort.
We collaborated with the client to develop a protocol and analysis plan.
Genesis Research Hub
Headquarters:
HOBOKEN
111 River Street, Suite 1120
Hoboken, New Jersey 07030, US